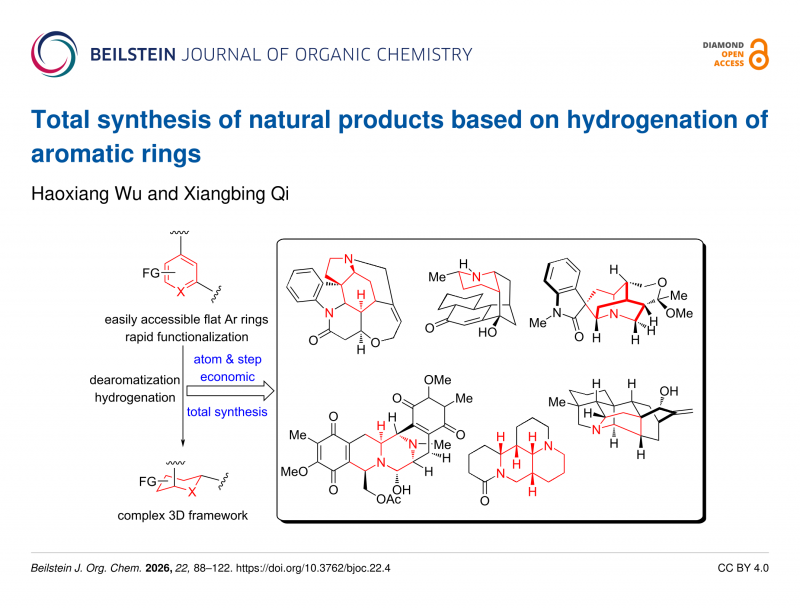
Total synthesis of natural products based on hydrogenation of aromatic rings
Haoxiang Wu and Xiangbing Qi
Arenes and heteroarenes are easily available building blocks in organic chemistry, and saturation the aromatic ring facilitates syn
thetic chemists to efficiently synthesize natural products with complex three-dimensional structures. Recent advances in catalyst
and ligand design have enabled unprecedented progress in the catalytic hydrogenation of (hetero)aromatic systems. Quinoline,
isoquinoline, pyridine, and related substrates can now be reduced with high efficiency and stereoselectivity, providing efficient
access to saturated and partially saturated architectures vital to synthetic chemistry. Furthermore, catalytic asymmetric aromatic
hydrogenation has facilitated the asymmetric total synthesis of complex natural products and pharmaceutical agents. This review
highlights recent advances in catalytic (hetero)arene hydrogenation, with a focus on its application in natural product synthesis.
http://qigroup.nibs.ac.cn/wp-content/uploads/2026/03/1860-5397-22-4.pdf