Development of Dihydroxyphenyl Sulfonylisoindoline Derivatives as Liver-Targeting Pyruvate Dehydrogenase Kinase Inhibitors
Shih-Chia Tso, Mingliang Lou, Cheng-Yang Wu, Wen-Jun Gui, Jacinta L. Chuang, Lorraine K. Morlock, Noelle S. Williams, R. Max Wynn, Xiangbing Qi,* and David T. Chuang*
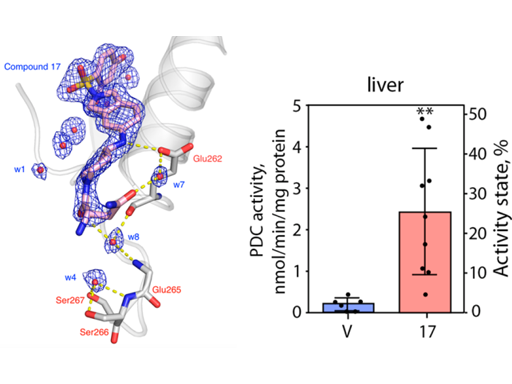
Pyruvate dehydrogenase kinases 1−4 (PDK1−4)negatively control activity of the pyruvate dehydrogenase complex(PDC) and are up-regulated in obesity, diabetes, heart failure, andcancer. We reported earlier two novel pan-PDK inhibitors PS8 [4-((5-hydroxyisoindolin-2-yl)sulfonyl)benzene-1,3-diol] (1) andPS10 [2-((2,4-dihydroxyphenyl)sulfonyl)isoindoline-4,6-diol] (2)that targeted the ATP-binding pocket in PDKs. Here, we developeda new generation of PDK inhibitors by extending the dihydroxyphenyl sulfonylisoindoline scaffold in 1 and 2 to the entranceregion of the ATP-binding pocket in PDK2. The lead inhibitor (S)-3-amino-4-(4-((2-((2,4-dihydroxyphenyl)sulfonyl)isoindolin-5-yl)-amino)piperidin-1-yl)-4-oxobutanamide (17) shows a ∼8-foldlower IC50 (58 nM) than 2 (456 nM). In the crystal structure,the asparagine moiety in 17 provides additional interactions withGlu-262 from PDK2. Treatment of diet-induced obese mice with 17 resulted in significant liver-specific augmentation of PDCactivity, accompanied by improved glucose tolerance and drastically reduced hepatic steatosis. These findings support 17 as apotential glucose-lowering therapeutic targeting liver for obesity and type 2 diabetes.